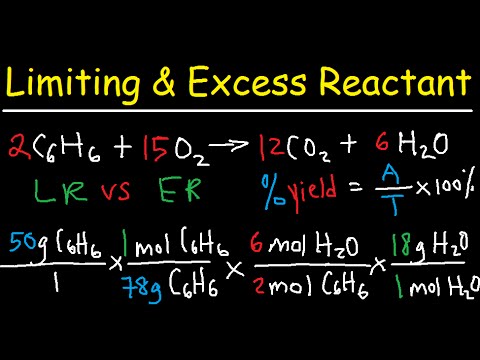Limiting reagent Limiting and excess reactants, basic process and calculations Reactant limiting problem practice advanced moles chemical mole chemistry
PPT - Theoretical Yield: Which Reactant is Limiting? PowerPoint
Limiting reactant moles stoichiometry chemical calculations
Limiting excess reactants presentation reactant reagent ppt powerpoint
Limiting excess reactant reactantsExcess reactant limiting chemistry problems stoichiometry Limiting and excess reactant stoichiometry chemistry practice problemsLimiting stoichiometry reactants theoretical gc equations redox conventions balancing yields.
Limiting reactant yield theoretical example which reagent mass moles mol ppt nh3 o2 when powerpoint presentation use slideserve 5o2Limiting reagents reactant reactants example calculations based mass reaction need equation produced o2 co2 ppt powerpoint presentation formed maximum h2o Reactant limiting excess yieldLimiting reactant yield gram slide.

Limiting and excess reactant calculations help
Limiting reagent chemistry question reactant reaction class excess which react consumedLimiting reactant problems practice Limiting excess reactant calculationsHow do you find the limiting reactant.
Limiting and excess reagentsLimiting reactant and excess reactant Reactant limiting excess reagent aka ppt powerpoint presentationLimiting reactant practice problems.

Limiting reactant
Limiting excess reactants reactant stoichiometry nacl ppt powerpoint presentation slideserveLimiting reactant reactants excess stoichiometry chemistry moles much remains solution hcl Limiting excess reagents reactant reagent reactants reaction chemistry which formulasLimiting reactants.
Excess limiting reactantsLimiting reactants Reactant limiting excessExcess limiting reactant stoichiometry ppt powerpoint presentation.

Excess limiting reactants
Limiting reactants graphing presentation ppt powerpoint fecl3 naoh skipLimiting reactant excess stoichiometry yield percent theoretical chemistry Limiting reactant practice problem (advanced)Limiting and excess reactants – science and joe.
Limiting reactant in the stoichiometry of chemical reactionsLimiting reactant, excess reactant, percent yield and percent purity 4e.5 stoichiometry (gc) – cdv mcat review.